While most consumers tend to think of probiotics – live microorganisms with proven health benefits – in relation to digestive or immune health, select probiotic bacteria have emerged as heart-health ingredients in recent years, ProBiotix Health CEO Steve Prescott told FoodNavigator-USA.
LP-LDL (Lactobacillus plantarum ECGC 13110402) – the bacterial strain developed by OptiBiotix Health – is unusual both in terms of its efficacy in the context of other food interventions known to lower LDL (from soy protein to oat beta-glucan and plant sterols), but also because of its blood pressure lowering effects, said Prescott.
Clear biomarkers, known mechanism of action
The mechanism of action is also well understood, he argued.
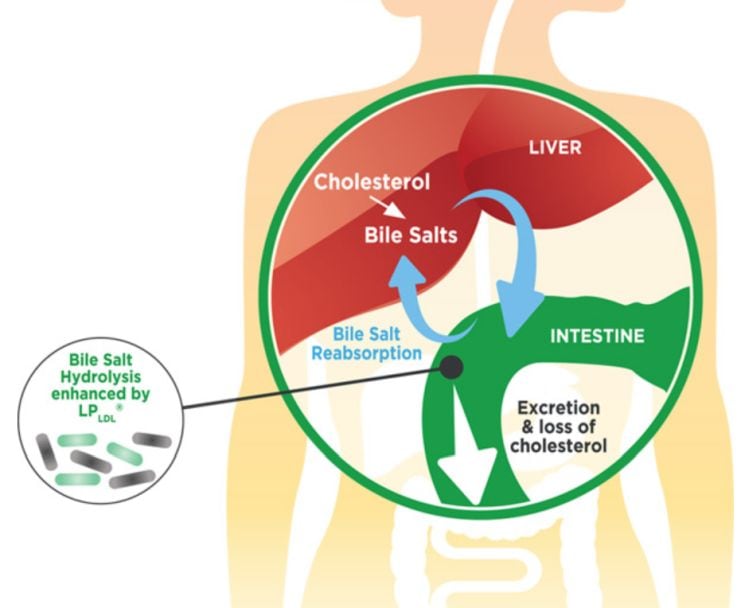
In a nutshell, the liver uses cholesterol in the blood to help it synthesize bile salts that help with the digestion of fats and other compounds. LP-LDL produces an enzyme called bile salt hydrolase that breaks down the bile salts in the gut such that they are excreted, prompting the liver to produce more bile salts, thereby taking more cholesterol from the blood.
“This is a known mechanism of action to reduce cholesterol, but very few bacteria produce enough of the enzyme to be clinically effective," he claimed. "There are only a few strains known to have enough enzyme activity to produce a clinical response and can work as a commercial product.”
There are competitive products on the market, said Prescott, including Lactobacillus reuteri NCIMB 30242 (LRC) from UAS Labs and a product from Kaneka called Floradapt Cardio that contains three strains of Lactobacillus plantarum. But he claimed: “Our overall LDL reduction is higher and we also have an indication for lowering blood pressure, which the other strains don’t have.”
Naturally occurring bacterial strain
The bacterial strain in LP-LDL [ECGC 13110402] is naturally occurring, said Prescott. However, OptioBiotix didn’t stumble across it and have a Eureka moment: “We screened first with a computer over 4,000 strains that might have potential, that led us to 150 targets that we took into the lab for more traditional screening, and then took those candidates into clinical."
The strain in question was selected as it demonstrated high in vitro bile salt hydrolase activity, cholesterol removal potential, resistance to gastric, pancreatic, and bile acids and high survivability on freeze dryingm he said.
“Unfortunately for a lot of things that probiotics are targeting like IBS, it’s about how you feel, but we’re always looking for a biomarker [ie. something that can be objectively measured - such as LDL cholesterol or systolic blood pressure – and is clearly associated with a health condition].”
The firm’s IP is around the ingredient and its applications in particular food products for cholesterol reduction, he said, whereas the proprietary screening techniques are not being patented.
The go-to-market strategy
Asked about the go-to-market strategy, he said: “We started around 15 months ago and started distribution in Europe and have since moved to the US and Asia, first with dietary supplements and have since moved into food, announcing a partnership with a dairy in Uruguay to launch a yogurt and a Brazilian company that produces a dry yogurt sachet for addition to fresh milk for making yogurt.”
He added: “If it’s in a very dry product, like a fiber or protein blend, LP-LDL is shelf-stable at room temperature, but if there’s moisture it needs to be refrigerated. Dairy is a really good vehicle, especially yogurt, but juices, chocolate and fresh milk are a bit more complicated; it has to be added post pasteurization.”
Right now, consumers don’t generally associate heart health with probiotics, so some education is needed, he said, but the appeal is clear given consumers are looking for natural ways to reduce cholesterol without unpleasant side effects.
“It’s not as strong as statins, but for a food intervention it’s at the higher end [for LDL-reduction], so it’s very exciting, particularly as companies are looking to add functionality to the dairy category.”
Human clinical data
So where’s the evidence that it works?
There is one published human clinical study funded by OptiBiotix Health in the peer-reviewed open access scientific journal PLoS One (Costabile et al, 2017), conducted at the University of Reading in the UK by scientists including Professor Glenn Gibson, who is internationally recognized for his research into pre- and probiotics.
A second, larger, human clinical study has just been completed in Italy that should be published this summer, said Prescott. A third human trial is being developed by the University of Roehampton, UK, although COVID-19 has delayed this.
The 2017 study was a double-blind, placebo-controlled, randomized human intervention study over 12 weeks with 49 normal to mildly hypercholesterolemic adults aged 30-65.
It found that daily ingestion of 2x109CFU (Colony-Forming Unit) encapsulated LP-LDL twice daily resulted in a statistically significant (13.9%) reduction in LDL cholesterol in volunteers with baseline total cholesterol of <5mM during the 0-12 week period and a 13.1% decrease in the group with more elevated LDL.
There was a 4.5% increase in HDL (‘good’) cholesterol for those taking the probiotic in the 6-12-week period, with a more meaningful (14.7%) increase for the over 60s.
‘A similar reduction to that of stanols and sterols may be achievable’
To put this into perspective, said the authors, the European Food Safety Authority (EFSA) reviewed 80+ studies and concluded that ingesting 1.5–2.4 grams a day of plant stanols/sterols can reduce LDL by 7-10.5%.
“The results of this study using Lactobacillus plantarum ECGC 13110402 suggest that a similar reduction to that of stanols and sterols may be achievable. These reductions have potential clinical significance as a 1% reduction in serum cholesterol has been associated with a lessening of artery disease risk by 2–3%.”
A statistically significant (5.1%) reduction in systolic blood pressure was also observed across the active study group in the 6-12-week period, noted the authors, although follow-up studies should include volunteers with hypertension, they added: “As this study was carried out in volunteers with normal blood pressure, the impact of the intervention should be confirmed in hypertension sufferers.”
They added: “Optimization of the study protocol, dosage timing, and targeting a higher cholesterol population and/or high blood pressure group and potential combination with a Lactobacillus plantarum targeted prebiotic would further enhance and demonstrate its cholesterol lowering activity.”
However, “further studies with larger numbers would allow a clearer representation of significant effects."
Health claims
While there are no hard and fast rules about how many clinical studies are needed to support claims or reassure customers, some companies are comfortable with one strong published study while others would like to see additional data, which should be coming fairly soon, said Prescott.
“However, the mechanism of action is accepted in the literature and the medical community, so we haven’t had too much pushback.”
As for on-pack claims, he said, while the clinical data shows a cholesterol-lowering effect, structure-function claims in the US typically require wording such as ‘supports healthy…’ or ‘helps maintain...' so firms likely would be looking to make claims such as ‘supports healthy cholesterol levels,’ he said.