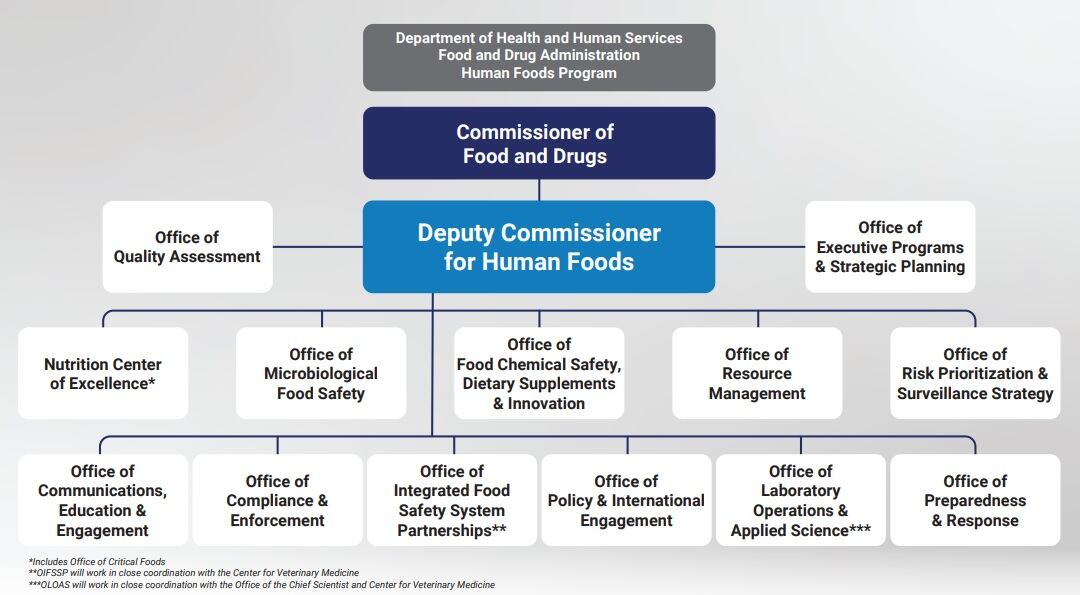
When Jones takes the hot seat on September 24, he will oversee 12 offices and one center and report directly to FDA Commissioner Robert Califf.
The proposed Human Foods Program would include food safety, chemical safety and innovative food products, including those from new agricultural technologies, as well as nutrition to help reduce diet-related diseases and improve health equity. One of the offices Jones would oversee is the newly proposed Office of Food Chemical Safety, Dietary Supplements & Innovation.
For more than 30 years, Jones has held various positions in the U.S. Environmental Protection Agency (EPA), stakeholder community and private industry around issues related to chemical safety and sustainability in the environment. His work has focused on lessening the impact that chemicals and pollution have on the U.S. food supply. At the EPA, he was a principal architect of the 2016 overhaul of the Toxic Substances Control Act, the first update of that statute in more than 40 years.
Notably, Jones was an integral member of the Reagan-Udall Foundation’s Independent Expert Panel for Foods, which submitted a report on the operational evaluation of the FDA’s Human Foods Program to the agency in December 2022.
“Our proposed reorganization is the largest undertaking of its kind in recent history for our agency,” said FDA Commissioner Robert Califf, MD. “I’m confident that under Jim’s leadership, we will build a stronger organization that will be integrated with other components of the FDA and focused on keeping the foods we regulate safe and nutritious, while ensuring the agency remains on the cutting edge of the latest advancements in food science and nutrition.”
“I had the pleasure of serving on the expert panel that provided operational recommendations for the FDA’s foods-related activities, and I now look forward to helping the agency realize its vision for the proposed Human Foods Program, including carrying out important nutrition initiatives to improve the health of our country,” said Jones.
“As a former pesticide regulator, I have a deep understanding of the unique needs of government programs involved in upholding safety of the U.S. food supply, as well as the important role that the agriculture community and state partners play in this paradigm. I am honored to serve the FDA and the country in this new capacity.”
CRN: “We are eager to share with him our concerns”
Jones’ appointment was met with hopeful welcome by Steve Mister, President and CEO of the Council for Responsible Nutrition (CRN).
“Today’s announcement reflects the FDA’s urgency to improve its oversight of food and strengthen consumer confidence in the safety of these products. We are hopeful Mr. Jones shares our interests in robust agency attention to dietary supplements as well,” said Mister.
“We congratulate Deputy Commissioner Jones and look forward to discussing the many challenges that face our industry, which includes the agency’s prioritization of our needs,” he added. “We are eager to share with him our concerns and the diverse needs of our membership, and to help him better understand the important space we occupy in the large category of products he now oversees.”
NPA: "There seems to be a knowledge gap when it comes to foods"
The Natural Products Association (NPA) called Jones’ selection "troubling".
“For more than 30 years, Mr. Jones has held various positions in the U.S. Environmental Protection Agency (EPA) while Mr. Jones is a known commodity in Washington D.C., it is unclear how his experience will benefit the foods program, there seems to be a knowledge gap when it comes to foods,” said Daniel Fabricant, Ph.D., president and CEO of the NPA.
“At a time where FDA foods program has a clear accountability-gap with Congress, industry, and Americans as demonstrated by the Reagan-Udall Report, which Mr. Jones was a panelist, how will a lack of technical understanding for the industries FDA regulates fill that gap. FDA needs to lay out constructive plans to address these deficiencies sooner rather than later. While the reorganization is significant undertaking it is not going to have a positive outcome if the new
commissioner for foods doesn’t engage immediately with groups like the Natural Products Association to understand the history and specific concerns.
CBA: "Jones enters the agency at a critical time"
Sarah Gallo, Vice President of Product Policy at the Consumer Brands Association, formerly the Grocery Manufacturers Association (GMA), said: “Jones embodies the management qualifications the industry and stakeholders have been asking for to ensure FDA can properly deliver on its mission of protecting consumer health and safety. We commend Commissioner Califf on this hire and are encouraged that this is a step toward modernizing the agency so it can move at the speed of the consumer.
“We are the makers of America’s food, beverage, household and personal care products that consumers depend on every day – providing products that are safe is our industry’s top priority. Our companies follow rigorous safety standards and comply with regulations set by the FDA. It is, therefore, imperative for the deputy commissioner to defend its existing and comprehensive science-based system to deter patchwork state regulations that put the burden on consumers and undermine public health.
“Jones enters the agency at a critical time for consumers and the industry with a number of important and impactful issues under consideration that deserve attention and engagement – from front-of-pack labeling to defining “healthy” to chemicals management. Consumer Brands is eager to partner with Jones to transform the agency into one that is structured, governed and funded for success.”
AHPA
Michael McGuffin, President of the American Herbal Products Association (AHPA), commented: “AHPA welcomes the selection of Jim Jones as Deputy Commissioner for Human Foods. We look forward to working with him as part of our ongoing engagement with FDA, and will communicate to him early in his tenure our continuing concern about the potential impacts of the proposed placement of ODSP under a larger office on the regulation of dietary supplements.”