SuperMeat is working on cultured chicken liver meat, minced meat and chicken breast meat
SuperMeat founder: ‘The first company that gets to market with cultured meat that is cost effective is going to change the world’

It also envisages a future whereby small businesses – and ultimately consumers - could produce small quantities of ‘meat’ locally in their own small bioreactors – using capsules containing ‘starter’ tissue produced by SuperMeat.
And unlike rivals in this embryonic field – who are focused on beef - SuperMeat is focused on cultured chicken (liver, minced, and breast meat), which could make its way into everything from chicken nuggets to strips, burgers and cutlets.
Indiegogo campaign is not just about raising money
The Tel Aviv-based company – which is seeking to raise $8.5m in the next six months or so – is led by Yaakov Nahmias, an acclaimed biomedical engineer and a professor of bioengineering at the Hebrew University of Jerusalem who is currently based at Harvard.
Professor Nahmias, a specialist in tissue engineering techniques refined from regenerative medicine, spoke to FoodNavigator-USA after SuperMeat launched a crowdfunding campaign on Indiegogo seeking to raise $100k.
While $100k won’t go very far considering the scope of the company’s ambitions (most of the funding for SuperMeat will come from different kinds of investors), the Indiegogo campaign was designed to prove that consumers – as well as foodtech investors - buy into the concept of cultured meat, said Nahmias.
“Indiegogo for us is a way to demonstrate that there is public support for what we’re doing. I think journalists often talk about the ‘yuck’ factor but I think there is an entire generation [of young people] out there that doesn’t think this is yucky at all.”
Conventional meat production – which involves the industrial scale slaughter of animals – is arguably considerably yuckier, not to mention environmentally unsustainable as the population continues to grow, he suggested, while it does not raise the same kinds of food safety issues (no more anxiety about antibiotics, salmonella and E.coli).
We use a mesenchymal type of cell that grows faster and in a cheaper medium
So what kinds of cells is SuperMeat using, and what inputs are required to help them grow and proliferate? These are commercially sensitive questions, said Nahmias, but he can say what kinds of cells he isn’t using.
“So like everyone else we start with a biopsy, so we take cells, but we don’t take muscle cells or muscle stem cells, which are satellite cells, we use a different type of cell – a mesenchymal type of cell that grows faster and in a cheaper medium as it has fewer nutritional requirements than myocytes [muscle cells] and satellite cells. Our cells have the unique potential of differentiating into myocytes and adipocytes [fat cells].
“They are uniquely capable of making muscle and fat which is very important for the taste of the meat. Really you can make so many things. One thing I’d love to make is foie gras, because the way they make it today is particularly cruel.”
As for the growth medium, he would not provide full details, but said: “So the culture medium needs to have certain growth factors and also hormones and nutrients like glucose and fatty acids.”
SuperMeat founder Professor Yaakov 'Koby' Nahmias is an award-winning biomedical engineer and the founding director of the Alexander Grass Center for Bioengineering at the Hebrew University of Jerusalem.
In 2006, his work established the first vascular liver tissue, which survived for over 50 days in the lab. The same year, he became a faculty member of Harvard Medical School, winning a National Institutes of Health (NIH) Career Award. In 2009, he secured two consecutive European Research Council (ERC) starting and consolidator grants, and won the Rappaport [biomedical research] Prize for his groundbreaking work on liver tissue engineering and the development of nanotechnology therapies for the treatment of diabetes.
SuperMeat does NOT use serum in its process
Unlike others in the field, said Nahmias, SuperMeat does not use animal serum (the liquid part of blood) in its process (some start-ups growing cultured beef, for example, are using fetal bovine serum, a byproduct of the livestock industry, which somewhat defeats the purpose of cultured meat).
He added: “Serum is cheap, but if you don’t use it you must tailor your culture medium to the nutritional and hormonal and growth requirements of cells; it’s complicated, but our cells are proliferating under very precise conditions.
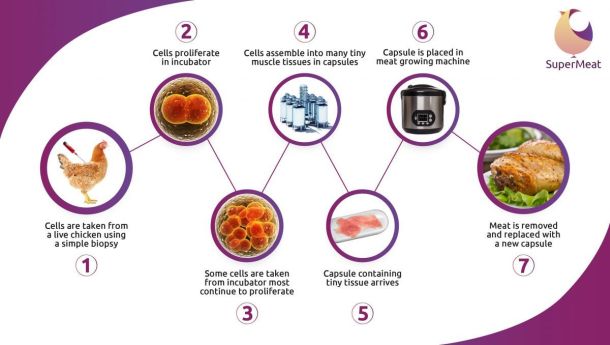
“Our process is unique. To start with, we take the cells and grow them like everyone else in bioreactors, but then the technology we use is different; we take very small amount of cells from the bioreactor and allow them to form tiny tissues – something like 14mg in weight or even smaller.
“The plan is that we’ll then take those tiny tissues and send them in small capsules to wherever people want to produce the meat locally [eg. to restaurants, food companies, and ultimately, even consumers] and then they will grow in small machines that enable them to grow from a few milligrams to a kilo or more of tissue inside the machine.”
How do you stop the cells from dying?
But why not make massive amounts of cultured meat centrally, and then distribute the finished product to end customers, as the regular meat industry does?
Because distributed, local production systems are the future, argued Nahmias, but also because of the nature of the tissue engineering process.
“The biggest problem in tissue engineering is the diffusion limits, because you can only get oxygen [which cells need to survive] about 200 microns into the tissue, and if your tissue is bigger than that then the cells in the middle will start to die from lack of oxygen/hypoxia.
“So if you want to build up muscle you need to do it under perfusion [a system enabling the oxygen and other nutrients to get to all the cells that need them], to continuously deliver nutrients to the cells like the animal body does via blood vessels.
“The tissue that we transport [to end customers/local ‘cultured meat’ producers] is small enough that it is below the 0.2mm limit that I mentioned. It has a fairly short shelf-life, but then so does [regular] chicken meat [from slaughtered animals].”
What’s incredible about the tiny tissues, he says, is that “they contain all the elements of the muscle and actually contain functional blood vessels that can interconnect to an existing perfusion and can grow and allow the entire tissue to grow with them.”
Local meat producer wouldn’t need expertise to operate the machine
As for the small machines - SuperMeat has come up with several prototypes but needs money to come up with something commercially viable. “The machine needs electricity, it needs to connect to the water supply, it needs heat, as it is an incubator, and end users will have to insert the capsule [of tissue] and nutrient powders.”
But no real expertize will be needed on the part of the end user, he stressed. “If it did, that would defeat the purpose.”
Game-changing technology
So where does he see the biggest commercial potential for the technology, and what are the milestones that lie ahead?
“Our aim is to have a public proof of principle tasting about two years from now. The first company that gets to market with a product that is cost effective is definitely going to change the world.”
As for the market potential from a regional perspective, this isn’t just technology for Europe and the US, although this is where the research is concentrated right now, he said.
“The biggest consumers of meat in the coming decade will be in Asia and I think people have seen the problems that industrial farming has created in the west and they will be looking for another way.”
As to the regulatory environment surrounding cultured meat, it’s new territory, but there are already conversations going taking place with the FDA and other agencies, he said. “Quite frankly, it should be fairly straightforward. It’s just meat.”