So the FDA and USDA will share oversight for cell-based meat… but what will this mean in practice?

Will pre-market approvals of the cell collection process, cell banks, and techniques for cell growth/differentiation be required - perhaps via the food additive petition process? Or will the GRAS process suffice (and is anything ‘generally recognized’ yet in the world of cell-based meat)? What might the inspection regime look like and precisely when does FDA jurisdiction end and USDA's begin? Where does seafood - which wasn’t mentioned in the joint statement - fit in? And what will we call cell-based meat on food labels?
Encouraging though it is to see the two agencies agree to work together, this is just the beginning of the process, Robert Hibbert, a partner at law firm Morgan Lewis, told FoodNavigator-USA.
“The November 16 announcement set some basic parameters, but all the difficult questions still have to be addressed and resolved.”
Memphis Meats: Give us some ‘bright lines’
According to the joint FDA/USDA statement, for example, “a transition from FDA to USDA oversight will occur during the cell harvest stage.”
But what precisely does this mean in practice?
In comments on the use of cellular agriculture to manufacture products derived from livestock and poultry stem cells, Eric Schulze, PhD, VP, product and regulation at cell-based meat start-up Memphis Meats, noted:
“As currently proposed, it is unclear when and how oversight would be transferred from FDA to USDA, particularly because the proposed framework calls for transfer of oversight ‘during the cell harvest stage,’ and this stage could involve multiple steps.
“We urge the agencies to identify a more specific point in the production process at which oversight would transfer from FDA to USDA, as doing so would help to provide ‘bright lines’ and a clear understanding of the roles and responsibilities of both agencies.”
Multiple and potentially overlapping FDA and USDA inspections?
As for how the facility inspection regime might work, he said, “Under the proposed joint framework, cell-based meat and poultry products would be subject to a dual-inspection regime, which could result in an individual product undergoing multiple and potentially overlapping FDA and USDA inspections.”
For example, when cell-based meat/poultry is used as an ingredient in multicomponent foods, the proposed division of responsibilities could result in an FDA inspection of the cell-based meat and poultry products in the pre-harvest stage, followed by inspection by FSIS (USDA) in the post-harvest stage, and then followed by inspection of the ingredient when used in a multicomponent food by FDA or FSIS, depending upon the amount of meat or poultry in the product, he speculated.
“This would mean that a cell-based meat or poultry product that will be used as an ingredient in a multi-component food, could be subject to at least three FDA or USDA inspections… It is imperative that the agencies clearly define their respective roles and responsibilities along with the specific regulatory requirements for each step in the process, to avoid confusion and inefficiencies.”
Interagency workgroup?
He added: “We propose that the agencies create an interagency workgroup on improving coordination regarding cell-based meat, poultry, and seafood products, with a focus on reducing overlapping inspection activities.”
"It needs to be emphasized that conventional meat and poultry products are contaminated with soil, animal waste, bacteria and other environmental contaminants. Accordingly, significant regulatory efforts are required to turn a high-risk food input into a low risk food output. This is not the case with cell cultured meat products which are effectively sterile at the point that they are further processed and packed into a consumer food product. Furthermore, the complete production process is characterized by full traceability and transparency.
"As a low-risk food, there may be no regulatory need for expanding USDA’s meat inspection services to cover cell grown meat products; effective mitigation of risks associated this level of food risk is currently effectively managed by the FDA."
Didier Toubia, CEO, Aleph Farms (Israeli cell-based meat start-up)
CSPI: FDA should require pre-market approval for cell based meat via a food additive petition process
As for the FDA's initial safety assessment, said Gregory Jaffe, biotechnology director at consumer advocacy group the Center for Science in the Public Interest (CSPI), it’s not clear whether the FDA is considering a GRAS-type process, a consultation process such as that used for new biotech plant varieties, or a food additive petition process (which requires pre-market approval).
The CSPI – which argues that “the absence of a mandatory pre-market approval process at FDA for genetically engineered crops has been a factor in the low consumer acceptance of those foods” – prefers the latter approach, he said:
“The FDA should require food additive petitions for novel ingredients, as GRAS notifications are inappropriate where a consensus among knowledgeable experts regarding the safety of an ingredient or input, by definition, does not yet exist. Absent further action from Congress, the FDA should utilize its food additive petition process, and allow public comment on such petitions, for every new condition of use for innovative and novel substances added to foods.
"Because the production of cell-based meat is certainly a new condition of use, the FDA must use the food additive petition process to ensure that every component that is used in processing and makes its way into the final product has been vetted for safety.”
Read all of the comments HERE.
What’s in a name?
Labeling was not referenced in the joint FDA/USDA statement of Nov 16, 2018, and remains a bone of contention, with advocates of the technology deploying neutral terms such as “cell-based meat” or “cell-cultured meat,” and others - notably the National Cattlemen’s Beef Association – choosing more loaded terms such as “lab grown fake meat.”
Niya Gupta, founder and CEO of cell-based meat start-up Forke & Goode, added: “We anticipate some type of industry moniker or logo in addition to the meat product label to signify the difference in production process, such as ‘grown directly from cells.’ ‘made using cells’ or ‘slaughter-free’ to clearly represent and communicate what we’re doing.”
Robert Hibbert, a partner at law firm Morgan Lewis, added that while food labeling regulations do not typically require companies to reference the production process if the end product is the same (citric acid is labeled as such if it's extracted from citrus fruits or made from feeding simple sugars to Aspergillus niger mold and then processing the resulting fermented compound using calcium hydroxide and sulfuric acid), "There is a broad consensus that at least as these products first come to market they need to be differentiated."
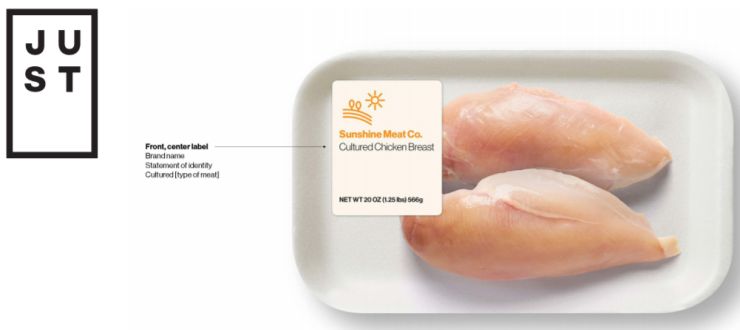
The picture above is a mock-up from JUST Inc, which is developing a variety of cell-based meat products: "We propose that labels for cultured meat products include the following elements: (1) On the front and center of the label: the name of the brand (e.g., “Sunshine Meat Co.”); and (2) just below the brand name: a statement of identity which indicates that the product is cultured, as well as the species from which the product is derived (e.g., “Cultured Chicken Breast”)."
What should we call cell-cultured meat? Click HERE.