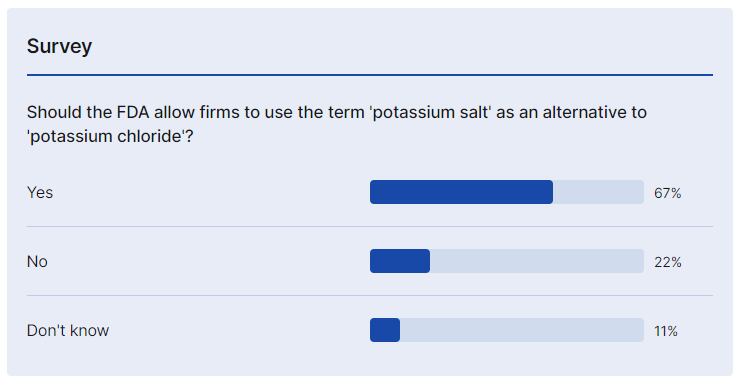
Petitioner NuTek Food Science - which has patented technology to tackle the bitter taste of potassium chloride, a popular salt replacer – argues that ‘potassium salt’ would ‘demystify’ the ingredient for consumers and help the industry achieve the federal government’s stated goals of lowering sodium, and increasing potassium intakes (from 3,500mg/day to 4,700mg/day).
Survey data shows that consumers believe potassium salt to be ‘more appetizing,’ ‘less processed,’ ‘safer to consume,’ and ‘healthier’ than the ‘chemical-sounding' potassium chloride, said NuTek president Brian Boor.
“A statistically significant number of consumers lack familiarity with the term potassium chloride and often mis-associate it with chlorine or other chemicals... ‘Potassium salt’ more closely reflects reasonable consumer expectations of the ingredient and more accurately describes the basic nature of the ingredient.”
CSPI: ‘We urge the FDA to give careful consideration to this petition’
In comments filed with the FDA, Washington DC-based health & consumer advocacy group the CSPI said it supported labeling that is “accurate and truthful, providing consumers with needed information to protect their health,” and that ‘potassium salt’ meets both of these criteria.

“Potassium chloride is a naturally occurring salt and is being used as a replacement for sodium chloride (which is commonly known as salt and labeled as such), providing many of the same properties sodium chloride does in food processing,” said president Michael Jacobson, PhD.
“In fact, insofar as the food industry can use potassium chloride to replace sodium chloride, it has the potential to reduce the population intake of sodium chloride. Current levels of sodium intake are a major public health problem in the United States.”
Unilever ‘fully supports’ NuTek petition: New name aligns with stated policy goals of FDA
Unilever also “fully supports” NuTek’s petition, said Patrizia Barone, PhD, regional regulatory affairs VP, North American Region and Global Foods & Refreshment, who argued that “recognizing ‘potassium salt’ as an alternate common or usual name would improve consumer understanding and promote clear food labeling.”
Unilever, she added, “has generated its own consumer data leading to similar conclusions. Unilever study results showed that consumer perception of ‘potassium salt’ was significantly better than potassium chloride…
"We ask FDA [to] grant the request in light of increased benefits to consumers and to help both the FDA and the food industry in our combined efforts to enhance the nutritional profiles of food.”
Salt Institute: Where will all this lead?
However, the Salt Institute - which represents the interests of salt producers - said that agreeing to a name change would open up a can of worms, given that scores of ingredients have 'chemical-sounding' names.
“FDA requires that vitamins be identified by the actual source of the nutrient (eg. 'sodium ascorbate' or 'pyridoxine hydrochloride') rather than by terms such as vitamin C or vitamin B6," said president Lori Roman.
“If ‘potassium salt’ is established as the common or usual name for potassium chloride, then presumably it would follow that ‘calcium salt’ would be the name for calcium chloride, ‘magnesium salt’ would be the name for magnesium chloride, and so on, when there is no evidence that those names are appropriate or would be understood.”

In a citizen’s petition filed with the FDA in June, NuTek president and COO Brian Boor argued that there is “compelling evidence that many consumers are confused and/or misled by the name potassium chloride… A statistically significant number of consumers lack familiarity with the term and often mis-associate it with chlorine or other chemicals."
He added: “Allowing entities to voluntarily label potassium chloride as ‘potassium salt’ encourages entities to make healthier food while demystifying potassium chloride for consumers. The proposed alternate name ‘potassium salt’ more closely reflects reasonable consumer expectations of the ingredient and more accurately describes the basic nature of the ingredient.”
Salt Institute: Consumers might think ‘potassium salt’ is a blend of table salt and potassium
The FDA has “long taken the view that minerals other than salt [sodium chloride] must be identified by specific common or usual names,” claimed Roman, who said the FDA had “appropriately denied” a citizen petition from the Corn Refiners Association requesting that companies be allowed to describe high fructose corn syrup as ‘corn sugar' [although the FDA did allow firms to call low erucic rapeseed oil 'canola oil' in the 1980s].
She added: “We are concerned consumers would be confused by 'potassium salt' and believe the ingredient is a form of conventional table salt or perhaps a blend of table salt and potassium."
A change in the common or usual name for potassium chloride would also require the FDA to conduct notice-and-comment rulemaking under the Administrative Procedure Act (APA), claimed Roman.
“Notice and comment rulemaking is necessary to allow the industry and other stakeholders to explore the full ramifications of the change.”
Sodium reduction
Their comments came as research published in the American Journal of Public Health revealed that the packaged food industry achieved a 6.8% reduction in sodium between 2009 and 2014.
The CSPI welcomed the news but said voluntary sodium reduction targets proposed by the FDA in June must be "quickly finalized" so that momentum is not lost.
- Read the comments on the NuTek petition HERE.
- Read the comments on the FDA's voluntary sodium reduction targets HERE.