In a mapping document penned by GFI senior scientists Liz Specht, Ph.D., and Christie Lagally, the authors note that, “Clean meat will likely find an early market entry point as a high-value ingredient in products that are predominantly plant-based.
“The first products that come to market may be hybrid products wherein clean meat is included as a part of plant-based products that essentially require only cell lines, media, and proliferative bioreactors to come to fruition.”
They add: “The next commercial products will likely be ground meat mimics, where scaffolding can be minimal; more complex structures requiring vascularization or perfusion bioreactors are not necessarily required.
“Finally, more structured tissues – like those mimicking steaks or chicken breasts – will require research and development in all of the areas outlined above. Thus, a consideration of target product(s) should drive the R&D focus.”
Speaking at a webinar hosted by the GFI on Wednesday, Dr Specht added: "I think there's a lot of evidence to suggest that consumers would be interested in hybrid products," citing the success of products already on the market that feature combinations of regular meat and plant-based ingredients, such as mushroom blend burgers and sausages with 40% meat, which are marketing on a health and sustainability platform, and can also be more affordable.
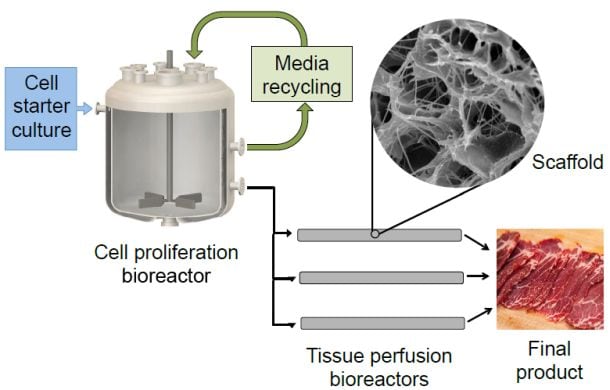
How do you make clean meat at scale?
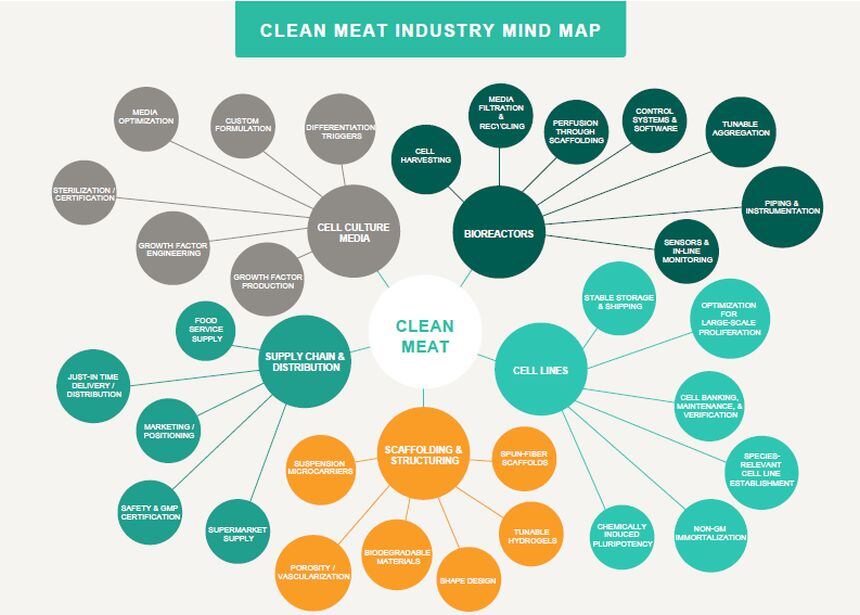
When you break the clean meat production process down into components – it requires cell lines (cells that will grow to make meat), growth media (food cells need to grow), and an initial bioreactor in which cells can proliferate.
But if you want to create products with a similar texture to steak or chicken breasts, you also need a second bioreactor (such as a tissue perfusion bioreactor) likely featuring some kind of scaffolding – which directs the differentiation of various cell types to encourage an organized pattern of muscle, fat, and connective tissue cells, says the GFI (a non-profit devoted to plant-based and clean meat and dairy alternatives).
“While the plant-based meat sector continues to make impressive strides toward parity with conventional meat in taste and texture, clean meat allows consumers to maintain their dietary preferences for animal meat while removing many of the inefficiencies and harms of current meat production.
“Clean meat production requires far less land and water than conventional meat, will produce exponentially less climate change, and eliminates the severe environmental repercussions of animal waste and contamination via runoff. It also requires no antibiotics, produces no bacterial contamination, and will not harm animals.”
Cell lines and growth media
Clean meat production begins with obtaining cell lines for the desired animal species (eg. chicken, pork, beef) that behave in a predictable way through many generations, while maintaining an unlimited capacity to reproduce/divide (ie. ‘immortalized’ cell lines).
In some cases, ‘primary cells’ directly isolated from an organism can spontaneously immortalize, says the GFI. However, more stable lines may be produced through genetic engineering whereby for example adult cells are reverted to stem cells, which can proliferate indefinitely and differentiate into multiple cell types (muscle, fat, connective tissue).
Meanwhile, other modifications or adaptations – such as the ability to grow in suspension, divide more quickly, or differentiate in response to unique environmental cues – may also be introduced in the cell lines.
Speaking to FoodNavigator-USA earlier this year, Dr Specht said: “Most companies in the clean meat space are working with cell lines that differentiate into all cell types, so they might work with mesenchymal stem cells for example, adult stem cells that can grow into muscle, fats, fibroblasts [connective tissues] and so on, that’s a common strategy.
“Another approach is to take a cell line - for example a fibroblast cell line - and do what is called trans-differentiation, so you take the cell line that’s differentiated into one of those [fibroblasts] and coax it to hop over to muscle or to fat.
“The challenge is to get the cells to differentiate into all those different cell types all at once, so simultaneously co-culturing those different cell types.”
"Meat is just a combination of cell types (muscle cells, fat cells, connective tissues), whether we're talking about a steak or fish or chicken. Essentially we are just growing these cells in culture and giving them a support structure to allows them to form into structures like meat in vitro as opposed to within an animal."
Dr Liz Specht, senior scientist, The Good Food Institute
Proliferation bioreactor
Next you have to proliferate cells, perhaps in a stir tank bioreactor where you might suspend your cells in the growth medium and stir them and keep them warm (there are already 20,000 liter stir tank bioreactors utilized in other markets). One approach is to use tiny beads as something for the cells to attach themselves to, which helps keep them suspended in the growth medium, but means you then have to find a way to harvest them (remove them from the beads) without killing them (eg. by adding enzymes that break down the attachments) and transfer them to a larger bioreactor to grow into meat.
Ideally, of course, you’d want to find a way to keep them suspended without having to be attached to anything, Dr Specht told us in December: "I see this stage as a continuous process where you might siphon off cells as they reach density, whereas stage two lends itself to more of a batch process.”
As for the growth medium (a nutrient solution containing salts, pH buffers, proteins, fats and growth factors), firms are looking for animal-serum free options, which are currently pretty costly, says GFI, although Memphis Meats told us in March that it had made significant progress on this front.
"All of the companies are moving away from using animal serum in their media," said Dr Specht. "There's a lot of room for making formulations far far cheaper, by for example using alternative production methods for growth factors."
Meanwhile, you’d also need media recycling systems to minimize inputs and waste, adds the GFI. “This system will require analytical sensors to measure concentrations of critical media components in real time and introduce fresh components as needed. Likewise, the system will need to be capable of filtering out waste products, maintaining pH and osmotic homeostasis, and maintaining real time quality control systems.”
Maturation bioreactor
As for the next stage, where you want to encourage the creation of an organized pattern of muscle, fat, and connective tissue cells, this would “probably require seeding onto scaffolding and then differentiating into the various cell types, the stage at which you get real fat cells forming and the muscle cells forming into fibers to give that authentic meaty texture,” said Dr Specht.
Here, the scaffolding would need to be something that is subsumed within the final meat product – so it would have to be made out of something that is degradable over time, or something edible that would not impact the taste or safety of the final product such as cellulose or collagen (there are animal-free versions in development – click HERE), she said.
“To accommodate three-dimensional growth, the scaffolds must exhibit porosity for perfusing nutrient media [the nutrients have to be able to reach the cells]. Alternatively, they must support vascularization of the tissue itself, i.e., the formation of a network of vessels to allow nutrients to permeate the tissue. Several production methods, including 3D printing and spun-fiber platforms, allow fine-tuning of pore size and microstructures within the scaffold.
"Once you get to the scaffold stage, that area is less explored and has not yet been demonstrated at scale."

"The phrase 'clean meat' is similar to 'clean energy' in that it immediately communicates important aspects of the technology—both the environmental benefits and the decrease in food-borne pathogens and drug residues."
Bruce Friedrich, executive director, The Good Food Institute (read more HERE )
Where next for clean meat?
Right now there are around half a dozen companies actively looking to commercialize clean meat, estimated Dr Specht, with scores more likely looking at it or working in areas that might service this burgeoning industry, she said.
Meanwhile, there is big potential for knowledge-sharing with related industries, from regenerative medicine and tissue engineering (eg. growing organs in vitro for transplantation); to stem cell therapy (where people culture human cells at a fairly large scale); antibody production (which uses large scale cell culture); and industrial biotechnology (things like enzyme production via large scale microbial fermentation).
In clean meat, as in any other emerging field, one of many challenges will be bridging the gap between academic research and private sector involvement, she said: "There's often a funding gap in the middle where you are taking things from proof of concept to a demo or pilot facility, and very promising technologies can sometimes drop off in this gap.
"So part of what we are going at the Good Food Institute is to look at unmet needs. Can existing companies fill gaps or should there be new companies?"
Read the GFI's mapping document.

Dr Liz Specht will give delegates at FOOD VISION USA 2017 the lowdown on clean meat, while Alex Lorestani will explain how Geltor is producing gelatin without animals. See the latest speaker list HERE.

San Leandro-based Memphis Meats, which unveiled a ‘beef’ meatball in February 2016 and cultured poultry meat in March 2017, has not gone into detail about the nature of the scale up process for producing its wares, but VP business development Steve Myrick told FoodNavigator-USA in March that production costs are continuing to go down and the company remains confident that it will have products on the market in 2021.
"We've learned a lot and we are continuing to reduce costs on a really rapid trajectory and we've had positive declines faster than we even expected to over the last several months."
While they may initially carry a premium price tag, Memphis Meats is confident that it will over time be able to produce meat at parity or below the price of conventionally-produced meat, in a manner that is greener, cleaner and kinder, said Myrick.
It has also made significant progress in validating an alternative to animal serum for the growth medium, the nutrient bath the cells need in order to survive and grow, he added.
"We've validated a production path that does not require serum, and we are in the process of rolling that out into everything we do."
(Fetal bovine serum - which pioneers in the clean meat space growing beef have typically used - is derived from blood extracted from a fetus after it is removed from a slaughtered dairy cow, which is not consistent with the philosophy underpinning cultured meat, and can also be inconsistent from batch to batch, and is in limited supply.)
The cells Memphis Meats is using come from adult stem cells or 'satellite' cells that are self-renewing, such that they will divide and grow indefinitely, said Myrick, who confirmed that the company is working with cells which can differentiate into all of the cell types necessary to make meat taste, look, cook and behave, like meat (eg. fat, muscle, connective tissue etc).
