FDA rethinks reorganization, allocates more authority to proposed deputy commissioner for human foods
Acknowledging “feedback provided by employees and stakeholders,” FDA Commissioner Robert Califf said yesterday the agency’s “thinking has significantly broadened” its proposal for a unified Human Foods Program to include expanded authority and a new model for the Office of Regulatory Affairs.
“We know that in front of us is a once-in-a-generation opportunity to unify our field work with the priorities of program offices and Centers. This is why I’m proposing a number of additional changes to ORA, including moving several of the office’s laboratories and merging its current compliance functions into those of the new HFP and other agency product Centers,” Califf explained in a statement.
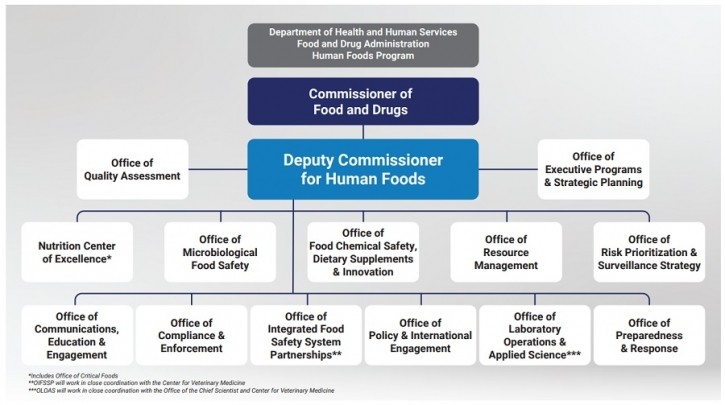
Under the revised proposal, ORA’s assignments for investigations, inspections and imports of all FDA-regulated products will be planned “in partnership” with the HFP and other product programs or centers, and compliance functions currently managed by ORA related to human foods will merge into the HFP to “streamline operations and expedite decision-making,” the agency said.
FDA also proposes to realign the eight human and animal food laboratories currently managed by ORA into the HFP and task the newly established and yet-to-be-hired Deputy Commissioner for Human Foods with working with the Chief Scientist and Center for Veterinary Medicine director to coordinate research.
Certain functions under the Office of Security and Emergency Management, will also move under the newly repositioned ORA, including the ability to activate Incident Management Groups to respond to emergency situations, such as recalls.
The agency also reiterated its earlier proposal to move certain aspects of international food safety and state and local food safety partnerships under an Office of Integrated Food Safety System Partnerships within the HFP.
Given the broad changes to ORA’s mission and structure, FDA may rename the office “to more appropriately align its title to the structure and functional duties of the agency’s field operations,” the agency said.
FDA responds to criticism
Many of these changes follow recommendations of an internal review of the agency’s botched response to an infant formula recall that resulted in massive national shortages and an external audit by the Reagan-Udall Foundation that sharply criticized many aspects of FDA’s culture and management structure.
Within six weeks of the release of the Reagan-Udall Foundation’s review, FDA announced plans for a unified Human Foods Program that earned accolades by many industry players and stakeholders, but also calls for additional action. Many felt that FDA’s self-proclaimed “transformative vision” for food safety and nutrition did not go far enough.
Common criticisms of earlier plans for the Human Food Program included that it didn’t pull together all components of the food group under one authority. For example, it left out parts of ORA related to inspection and compliance, food-related laboratories, import oversight, state partnerships, training and information technology. An oversight some argued was a “half step” approach that would leave the food program fragmented and unable to address much-needed cultural changes.
‘Bolder action’ earns FDA accolades
With yesterday’s announcement, however, some stakeholders are more optimistic about the agency’s future.
“We are pleased the FDA is taking bolder action to make meaningful and lasting change by answering informed industry and stakeholder calls to unite and elevate the Human Foods Program and fully authorize the deputy commissioner with control over its strategic direction. This includes complete authority over the budget, as well as responsibility to execute a common mission throughout al components, including the animal foods program,” Consumer Brands Association VP of Product Policy Sarah Gallo said in statement.
She added the trade group was “also encouraged that the FDA plans to address areas of duplication and refocus the Office of Regulatory Affairs (ORA) on its core business, inspections and investigations.”
While Gallo said CBA sees yesterday’s revision of the proposed reorganization as a “necessary step toward transforming the agency into one that is properly equipped to ensure consumer safety and meet rapidly evolving consumer demand,” others may see the agency as still falling short of its mandate.
A potential flashpoint is the proposal that ORA’s core mission will focus on conducting investigations as well as inspections – which suggests a reactive approach rather than a preventive one.
A new leader could be named soon
While FDA continues to work out the kinks for the full scope of authority and structure of the propose Human Foods Program, it could be nearing the end of a far-reaching recruitment effort to fill key positions.
The agency said yesterday it is in the “final stages the recruitment process for the Deputy Commissioner for Human Foods and will be providing an update in the near future.”
It also reiterated it is “on target” to finalize its reorganization this fall.










